|
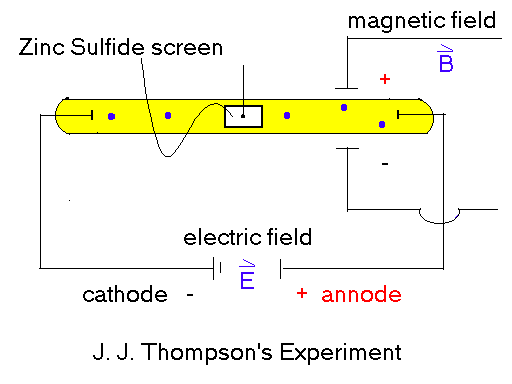
figure
1.1.3
This
same effect can be seen today if one were to bring a sufficiently
strong enough B field close to the sides of a television
screen. The electrons' trajectory will be changed as
this field is applied. From these experiments, the view
of the electron changed from the ancient's view to a more
modern view. Figure 1.1.4
gives us a picture of the Thomson model of the atom.
Thomson received the Nobel Prize in 1906.
|